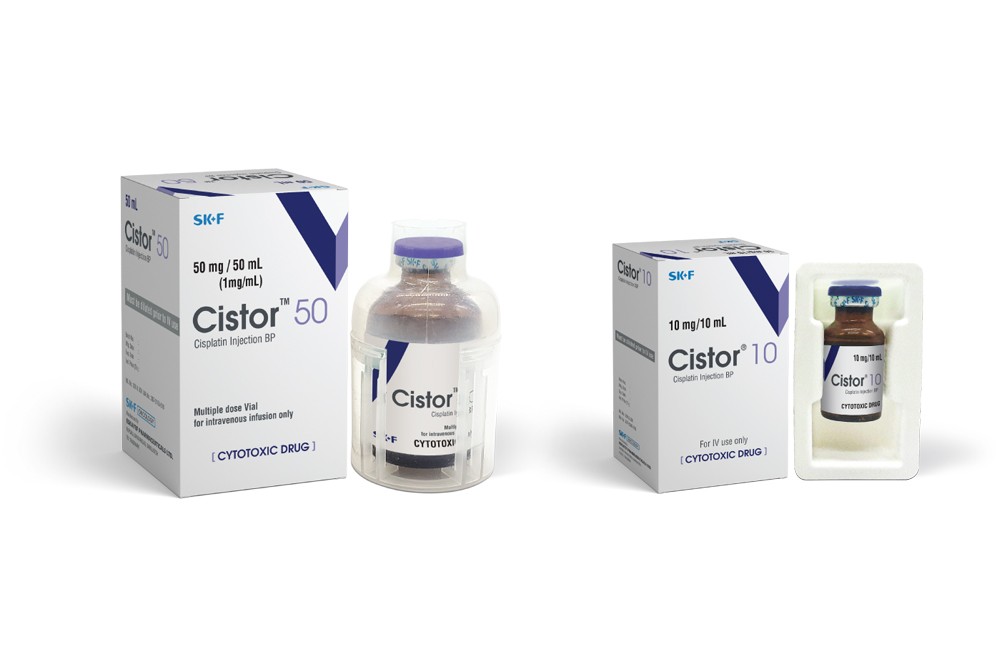
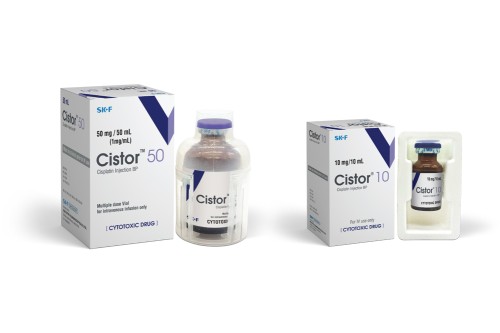
Indications
Cistor is indicated as first line therapy for lung cancer, head & neck cancer and for the treatment of advanced and metastatic ovarian carcinoma, bladder carcinoma and testis carcinoma.
Pharmacology
Cisplatin modifies cell cycle by interfering with DNA structure and function. Effects are most prominent during the S phase but cells are killed at all stages. Cisplatin synergises with other anticancer drugs e.g. fluorouracil. It has a narrow therapeutic margin and is highly toxic.
Dosage & Administration
Metastatic ovarian cancer: As monotherapy: 100 mg/m2 per cycle, given as a single dose infused in 0.9% sodium chloride or glucose once every 4 wk. For combination therapy with cyclophosphamide: 75-100 mg/m2 on day 1 of every 4-wk cycle.
Metastatic testicular tumours: 20 mg/m2 BSA daily for 5 days per cycle.
Advanced bladder cancer: 50-70 mg/m2 per cycle once every 3-4 wk, depending on the extent of prior exposure to radiation and/or chemotherapy treatment. An initial dose of 50 mg/m2 every 4 wk may be used in heavily pre-treated patients.
Usual dosage and schedule: 40 to 120 mg/m2 I.V. on day 1 as infusion every 3 weeks. 15 to 20 mg/m2 I.V. on days 1 to 5 as infusion every 3 to 4 weeks. The usual dose in adults and children when used as single agent therapy is 50-100 mg/m2.
Interaction
Synergistic with 5-fluorouracil and etoposide. Efficacy increased and toxicity reduced when combined with radioprotecting agent WR 2721. At doses ≤100 mg, Cistor is an ideal drug to combine with other cytotoxic drugs; unlike other antineoplastic drugs, it causes little myelosuppression.
Contraindications
Cisplatin is contraindicated in patients with pre-existing renal impairment. Cisplatin should not be employed in myelosuppressed patients, or patients with hearing impairment. It is also contraindicated in patients with a history of allergic reactions to cisplatin or other platinum-containing compounds.
Side Effects
Severe nausea and vomiting. Serious toxic effects on the kidneys, bone marrows and ears. Hypomagnesaemia, hypocalcaemia, hyperuricaemia. Peripheral neuropathies, papilloedema, optic neuritis, seizures. Ototoxicity (children) manifested as tinnitus, loss of hearing, deafness or vestibular toxicity.
Pregnancy & Lactation
Pregnancy Category D. There is positive evidence of human foetal risk, but the benefits from use in pregnant women may be acceptable despite the risk (e.g., if the drug is needed in a life-threatening situation or for a serious disease for which safer drugs cannot be used or are ineffective).
Precautions & Warnings
Patients with renal or hepatic disorder, myelosuppression. Monitor renal, neurological and auditory function. Perform blood counts regularly. Maintain adequate hydration before and 24 hr after admin to minimise nephrotoxicity.
Overdose Effects
Acute overdosage may result in kidney failure, liver failure, deafness, ocular toxicity, significant myelosuppression, intractable nausea and vomiting and/or neuritis. Death may also occur following overdosage. Treatment should include general supportive measures.
Reconstitution
Cistor lyophilized powder for injection 10 mg and 50 mg should be reconstituted with 10 ml and 50 ml of water for inj. respectively.
Cistor solution after reconstitution should be added to 2 liters of 5% glucose or 0.9% saline and intravenously administered in 6-8 hours; after administration an adequate hydration and diuresis should be maintained during 24 hours.
Storage Conditions
Stability: The Cistor remaining in vial following initial entry is stable for 28 days protected from light or for 7 days under fluorescent room light.
Storage: Product should be stored at controlled room temperature 25° C and protected from light.